eNose in malignant mesothelioma – Prediction of response to immune checkpoint inhibitor treatment
Aim: To assess as proof of concept whether the eNose was able to discriminate at baseline between mesothelioma patients with and without clinical response to anti-PD-1 plus anti-CTLA-4 therapy.
Take home message: Exhaled breath analysis using eNose technology can effectively differentiate between responders and non-responders to immune checkpoint inhibitors in patients with malignant pleural mesothelioma. This non-invasive approach offers a promising tool for predicting treatment outcomes, potentially enabling personalized therapy and avoiding ineffective treatments.
Introduction
Malignant pleural mesothelioma (MPM) is a rare cancer mainly caused by asbestos exposure. Despite advances in immune checkpoint inhibitors (ICIs), which show promise in improving outcomes, only a subset of patients benefit from this treatment. Identifying responders upfront could optimize patient care. This study evaluated the use of exhaled breath analysis with an electronic nose (eNose) to differentiate between responders and non-responders to ICI therapy (nivolumab and ipilimumab) in MPM patients.
Methods
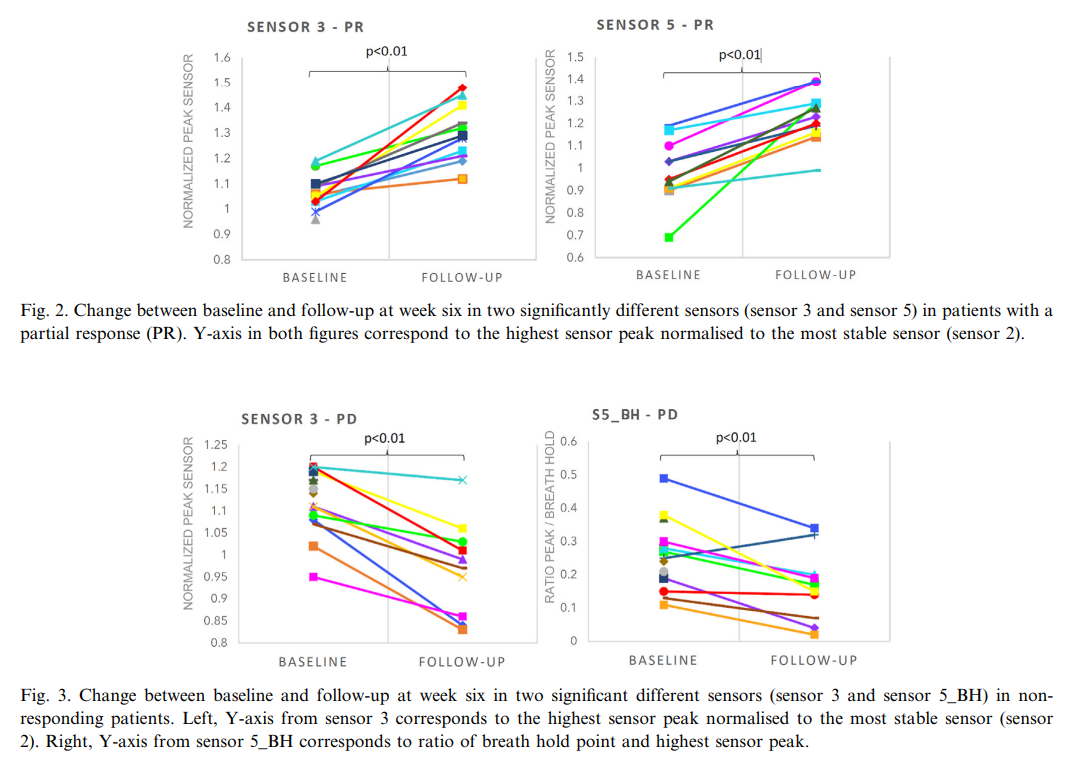
A prospective observational study was conducted as part of the INITIATE trial, which included patients with recurrent MPM treated with a combination of nivolumab (anti-PD-1) and ipilimumab (anti-CTLA-4). Exhaled breath samples were collected using the SpiroNose® at baseline and after six weeks of treatment. Breath profiles were analyzed in real-time on the BreathBase® platform. Response to treatment was assessed at six months using modified RECIST criteria. Patients with complete response (CR), partial response (PR), or stable disease (SD) were classified as responders, while those with progressive disease (PD) were classified as non-responders.
Results
The study included 31 patients, of whom 52% were responders (16 patients), and 48% were non-responders (15 patients). Baseline breath profiles showed significant differences between the two groups, with a cross-validation accuracy of 71% and an area under the ROC curve (AUC) of 0.90 (95% CI: 0.80–1.00). Follow-up breath analysis after six weeks indicated significant changes in sensor values for both responders and non-responders, but in opposite directions. Responders showed increased sensor values, while non-responders showed a decrease. These differences highlight the potential of eNose technology as a predictive tool for treatment response.