Exhaled breath profiles to detect lung infection with Staphylococcus aureus in children with cystic fibrosis
Aim:
- To investigate whether the eNose can be used to detect Staphylococcus aureus airway infection in children with CF.
- To explore whether CFTR modulator therapy with drugs that corrects the underlying defect of CF, alters exhaled breath profiles in treated children with CF.
Take home message: ENose technology offers a promising, non-invasive way to detect Staphylococcus aureus and other infections in children with cystic fibrosis. This rapid breath analysis could improve early diagnosis and monitoring, making infection management easier and more accessible in routine clinical settings.
Introduction
This study explores the use of electronic nose (eNose) technology to detect lung infections caused by Staphylococcus aureus (SA) in children with cystic fibrosis (CF), aiming to provide a non-invasive, real-time diagnostic tool for clinical use. Traditional CF pathogen detection relies on invasive or difficult-to-obtain samples, especially challenging for young patients and those on CFTR modulator therapy. This research evaluates eNose technology’s potential to serve as a point-of-care diagnostic alternative.
Methods
The study enrolled 100 children with CF (median age: 12 years) who were clinically stable and attending regular visits at a pediatric clinic. Using the SpiroNose®, patients provided exhaled breath samples that were then analyzed on the BreathBase® platform, which compared them to VOCs (volatile organic compounds) profiles from 25 healthy controls. Each child’s breath profile was processed to capture unique patterns associated with SA and other CF pathogens, using linear discriminant and ROC analyses to assess diagnostic accuracy.
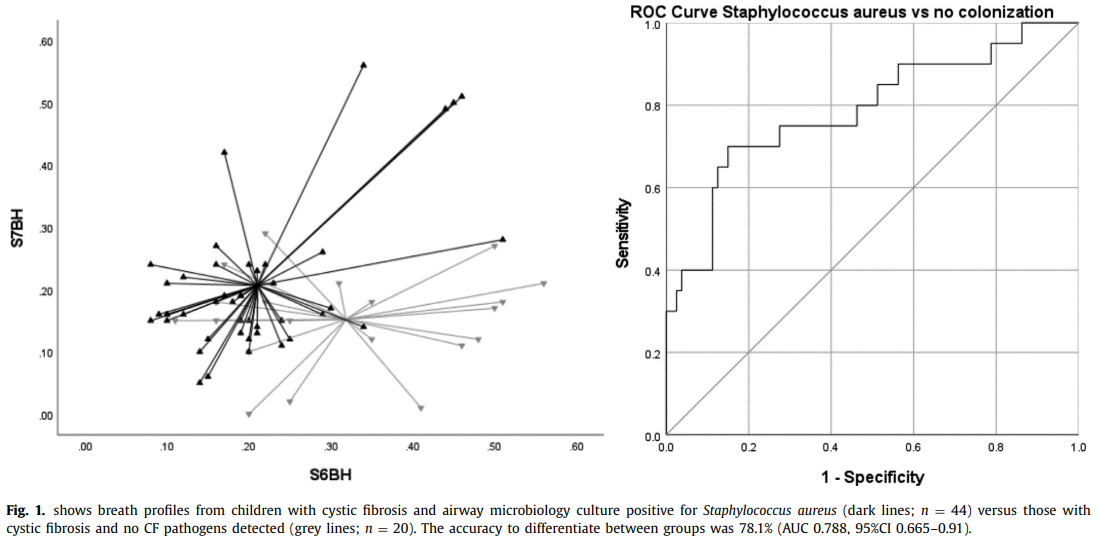
Results
The eNose successfully differentiated CF patients with SA from those without pathogens, achieving an accuracy of 74% (AUC-ROC 0.797) and effectively identifying CF patients with Pseudomonas aeruginosa (PA) as well, showing 78% accuracy (AUC-ROC 0.876). Additionally, CF patients with both SA and PA showed distinct profiles, suggesting pathogen-specific breath signatures. The study found no significant differences between breath profiles collected by throat swabs or sputum cultures, underscoring eNose technology’s robustness compared to conventional methods.