Prediction of response to anti-PD-1 therapy in patients with non-small-cell lung cancer by electronic nose analysis of exhaled breath
Aim: To determine the accuracy of exhaled breath analysis at baseline for assessing nonresponders versus responders to anti-PD-1 therapy in NSCLC patients.
Take home message: Exhaled breath analysis using electronic nose (eNose) technology can effectively identify distinct clinical and inflammatory phenotypes in patients with chronic airway diseases like asthma and COPD, regardless of their diagnostic classification. This approach captures underlying inflammatory processes through VOC patterns, providing a potential tool for personalized diagnosis and targeted treatment strategies beyond conventional disease labels.
Introduction
Immune checkpoint inhibitors (ICIs) have revolutionized treatment for advanced non-small-cell lung cancer (NSCLC), yet many patients do not respond effectively. Current biomarkers, such as PD-L1 expression assessed through immunohistochemistry, have limited predictive value. This study explores an innovative, non-invasive approach using an electronic nose (eNose) to analyze exhaled breath for predicting response to anti-PD-1 therapy. The hypothesis is that breath profiles can capture underlying metabolic and inflammatory changes linked to patient responsiveness, providing a faster and less invasive alternative to existing methods.
Methods
Study Population: The study included patients with advanced NSCLC eligible for anti-PD-1 therapy (nivolumab or pembrolizumab). It was a prospective observational study with two cohorts: a training set (March 2016 – April 2017) and a validation set (after April 2017). Patients were excluded if they had recent alcohol intake or were unable to participate.
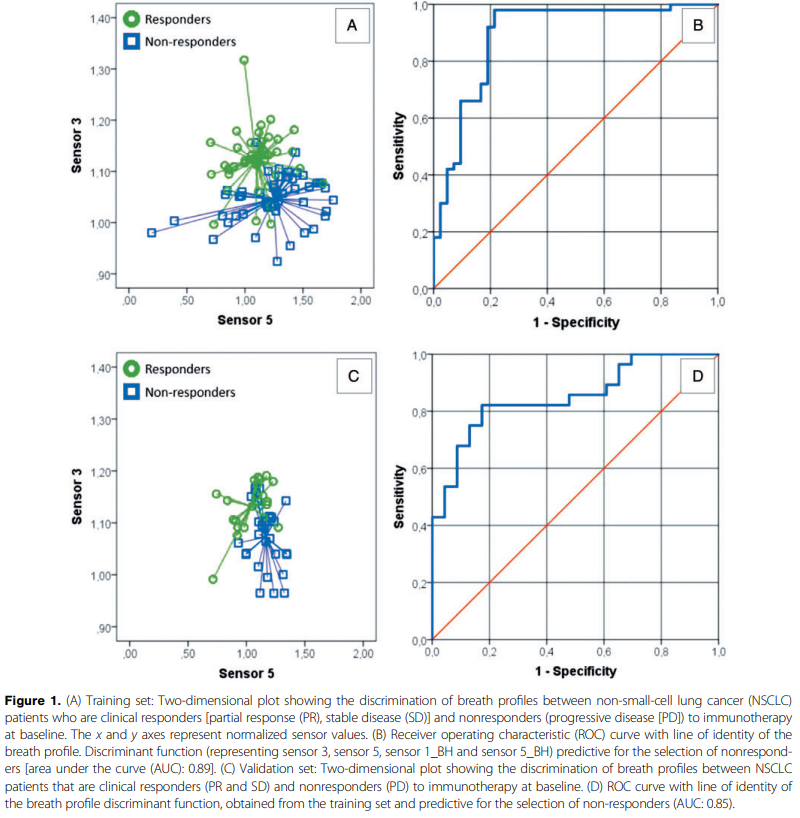
Measurements: Breath samples were collected using the SpiroNose® prior to the start of anti-PD-1 therapy (nivolumab or pembrolizumab). Exhaled breath was analyzed using seven cross-reactive sensors, and the signals were corrected for ambient air. Data were transmitted in real-time to the BreathBase® server for storage and analysis. The response to therapy was evaluated at 3 months using the RECIST 1.1 criteria, categorizing patients as responders (partial response/stable disease) or nonresponders (progressive disease).
Data Analysis: The eNose sensor data underwent pre-processing (filtering, detrending, ambient correction), followed by statistical analysis using independent t-tests, linear discriminant analysis, and receiver operating characteristic (ROC) curves. Predictive models were developed in the training set and validated in an independent cohort.
Results
The study analyzed breath samples from 143 patients (92 in the training set and 51 in the validation set). Significant differences were observed in the eNose sensor readings between responders and nonresponders. The model developed from the training set showed high predictive accuracy, with an area under the curve (AUC) of 0.89. In the independent validation set, the AUC was confirmed at 0.85, demonstrating the robustness of the eNose prediction. A cut-off point was established to identify nonresponders with 100% specificity, achieving a sensitivity of 43%. This approach outperformed PD-L1 IHC, which showed a lower AUC of 0.66.